Description
- Powerful antioxidant against free radical damage
- May help normalize blood glucose levels
- May support reducing anxiety and seizures by modulating GABA receptors
- May help activate PPAR, which has been clinically shown to be helpful in treating Alzheimer’s disease (AD)
- May be a safe adjunctive therapy during cancer treatments
Zetpil Suppository Advantage
Zetpil™ delivers nutrition via the rectum with the use of suppositories that have a unique, all- natural formulated base that has revolutionized not only this mode of delivery, but also the ability to obtain nutrients in a far more efficacious method. These suppositories are a result of combining natural ingredients with a non-irritating, rapid melting (5-6 minutes), readily absorbable (superior retention), antioxidant base material (free radical scavenging ability) and delivers the nutrients directly into the bloodstream in less than 20 minutes in a highly effective manner. Oral supplements are exposed to the hazards of stomach acid, digestive enzymes, food-related breakdown, first pass metabolism, and other bioavailability issues in the gastro-intestinal system.
Additionally, many raw materials simply have a very low bioavailability , and only achieve around 10% or less (commonly <1%) absorption rates, even if the patient is in optimal health condition. Compromised health, age, or certain conditions that result in an inability to take oral nutrients often preclude these patient populations from obtaining the necessary nutrition to stay healthy which often exacerbates chronic health issues.
Zetpil™ suppositories are a NOW solution to delivering the necessary nutrients to patients, on a daily basis, that is both easy, comfortable and cost effective. The safety and noticeable effectiveness of these products will far outweigh any initial hesitation you have regarding this mode of delivery.
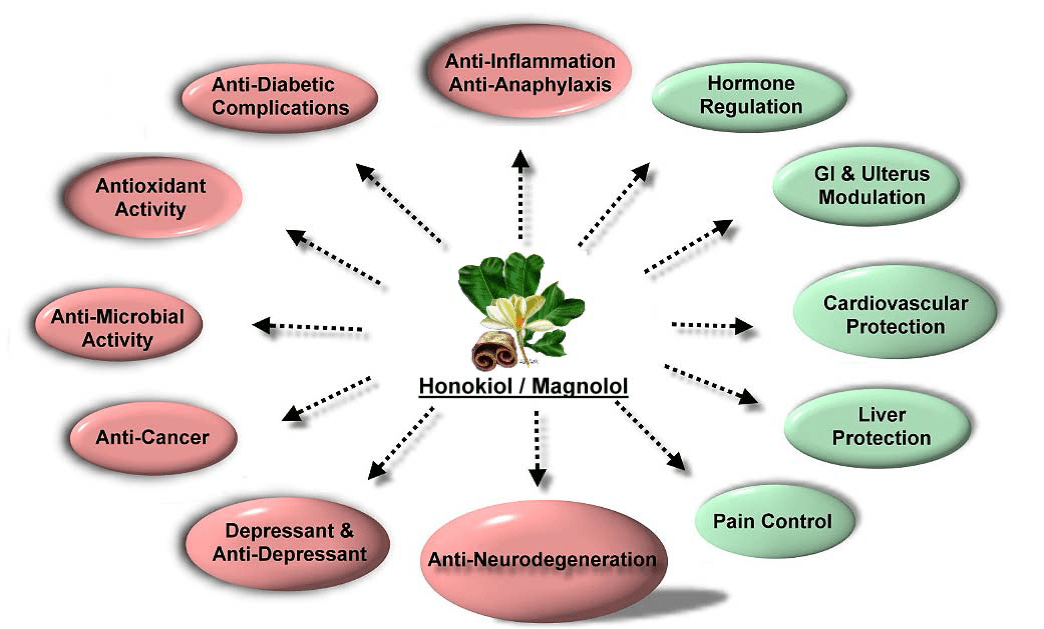
What is Honokiol?
Honokiol is a small molecule polyphenol from Magnolia grandiflora which has been shown to have antioxidative, anti-inflammatory, anti-tumor, anti-diabetic, anti-microbial, anti-neurodegeneration, and anti-depressant properties. Honokiol easily moves across the blood brain barrier making it an attractive compound for brain cancers and neurodegenerative diseases.
Honokiol has also been shown to have potent analgesic (pain relief) properties. Its anti-inflammatory properties have shown tremendous benefit for inflammatory disorders like rheumatoid arthritis, psoriasis, inflammatory bowel disease and asthma. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2765942/
Honokiol’s Therapeutic Properties
Anxiety Disorders
“Many drugs used clinically to treat anxiety, insomnia, and seizures act by enhancing GABAergic neurotransmission, and honokiol has been shown to bind to and positively modulate GABAA receptors.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3652012/
Diabetes
“In spite of being effective in normalization of blood glucose levels (Type II Diabetes), the currently used PPARγ agonists from the thiazolidinedione type have serious side effects, making the discovery of novel ligands highly relevant. Honokiol has been demonstrated to improve blood glucose levels and reduce weight gain in diabetic animal models.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4212005/
Neurodegeneration
“It is our view that PPAR activation remains a promising avenue for the treatment for Alzheimer’s disease (AD). However, the poor blood brain barrier (BBB) permeability of the currently available agonists and the negative outcome of the phase III clinical trials are likely to diminish interest in pursuing this target.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3156324/
Yet, we have safe natural compounds (ie: Honokiol) which have proven their effectiveness in clinical research to be able to effectively modulate the PPAR pathway. As there are no currently effective pharmacological treatments for the prevention and/or treatment of Alzheimer’s disease, Honokiol may represent a promising new therapy to reduce the consequences of neurodegenerative diseases.
Cancer
“Honokiol has been shown to reverse the multidrug resistance gene, and perhaps the ABCC transporter gene, the two major mechanisms for drug efflux. The (other) major role of honokiol may be facilitating the efficacy of conventional chemotherapy, by inhibiting NF-kB.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2842137/
“The use of natural products has been a successful approach towards managing (cancer), and the present review discusses the accumulating literature on the anti-tumor activity of honokiol against various malignancies. Honokiol may:
- inhibit the production of numerous cytokines, inducible enzymes, and adhesion molecules associated with tumor initiation, promotion, progression, metastasis and therapy-resistance.
- target chemopreventive and therapeutic pathways, such as NF-κB, STAT3, EGFR, mTOR and MAPK.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3663139/
HTTP://WWW.NCBI.NLM.NIH.GOV/PUBMED/20877235
Research shows Rectal Delivery Superior to Oral Delivery of Honokiol
Rectal delivery may achieve higher systemic dosages and may have a greater probability of reaching research-proven therapeutic dosages. When low oral bioavailability is the main hurdle to the clinical utilization of powerful natural compounds, suppositories may offer a potential solution.
“The maximal plasma concentration of honokiol following rectal administration (13.5 mg/kg of honokiol) was approximately six times to that administered orally at an identical dose indicating that rectal dosing avoids first-pass metabolism.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3663139/
Supplement Facts
|
Serving Size: 1 suppository
Servings per Container: 30 |
|
|
Each Honokiol 300 SR (Sustained Release) contains the following ingredients:
300 mgs in a blend of a lower concentration 2-(4-hydroxy-3-prop-2-enyl-phenyl)- 4-prop-2-enyl-phenol and the purest and highest concentration of 98% 2-(4-hydroxy-3-prop-2-enyl-phenyl)- 4-prop-2-enyl-phenol from the bark Magnolia grandiflora tree. This blend was created to insure both comfort as well as efficacy. |
|
| Suppository Base: The base is a proprietary combination of plant and fruit butters, medium chain triglycerides, phospholipids, lecithin and arrow root starch. | |
This product is contraindicated in individuals with a known hypersensitivity to phospholipids, or any of the listed ingredients.
If you choose to use this product as a rectal suppository, it is highly recommended you attempt to clear your bowels prior to inserting it to avoid premature evacuation. To facilitate insertion it is recommended you moisten the product by passing it under a stream of cool water or use a personal lubricant. When used as a suppository, this product requires 15-20 minutes retention to ensure the complete absorption of the bioactive ingredients. This suppository can be inserted in any position. Although not required, if inserted while lying on one’s side, consider remaining in the supine position for 1-3 minutes following insertion. (Always insert flat end of suppository first).
WARNINGS:
- Although safe for children, this product should be kept out of the reach of children.
- Do not use if the suppositories are open or damaged.
- As with any nutrient if you are pregnant or breastfeeding, seek the advice of a health care provider before using this product.
Individuals that have chosen to take this product as part of a personal nutrient program have overwhelmingly chosen to utilize this product as a rectal suppository. However, this product can also be taken safely by mouth. If one chooses to take this product orally, then this product may be considered a dietary supplement in accordance with current FDA guidelines.
Disclaimer: These statements have not been evaluated by the FDA. This product is a Federal Food, Drug, and Cosmetic Act compliant combination of naturally derived FDA GRAS (Generally regarded As Safe) herbal, nutraceutical, vitamins, minerals and amino acids products legally sold in the USA. In accordance with FDA regulations, when taken orally, this product may be considered a dietary supplement as defined by section 201(ff) of the Act, 21 U.S.C. § 321(ff).
Also, this product consists ONLY of a combination of naturally derived FDA GRAS (Generally regarded As Safe) herbal, nutraceutical, vitamins, minerals and amino acids products legally sold in the USA. Therefore, this product cannot be considered a drug as defined in section 201(g)(1) of the Act, 21 U.S.C. § 321(g)(1). This product is not intended to affect the structure or function of the body and is not intended for use in the diagnosis, cure, mitigation, treatment or prevention of disease in man. Any nutritional suggestions and research regarding this product are provided for informational purposes only, and are not intended to diagnose, treat, cure or prevent disease nor should any information provided be used as a substitute for sound medical advice.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.